Research
Mutations of tyrosine kinase receptors
We have developed and applied a bioinformatic tool to analyze pan-cancer missense mutations at the protein domain level. This approach allows to pinpoint novel rare cancer-associated variants and to highlight their correspondence with well-known mutations. We are characterizing the role of various mutations of the tyrosine kinase domain of tyrosine kinase receptors in cancer progression and response to therapies. Our studies could help to predict the inter-individual differences in terms of prognosis and response to drugs, improving the application of precision oncology.
Obesogenic behavior and climate change
We have recently started a new research project focused on understanding how metabolic features and lifestyle of obese individuals affect adverse climate change. In collaboration with Discovery foundation (Greece) company, we are investigating how obesogenic behaviors, including the consequent metabolic adaptations, can contribute to altering the environment. Our studies will likely pinpoint novel strategies to preserve our world.
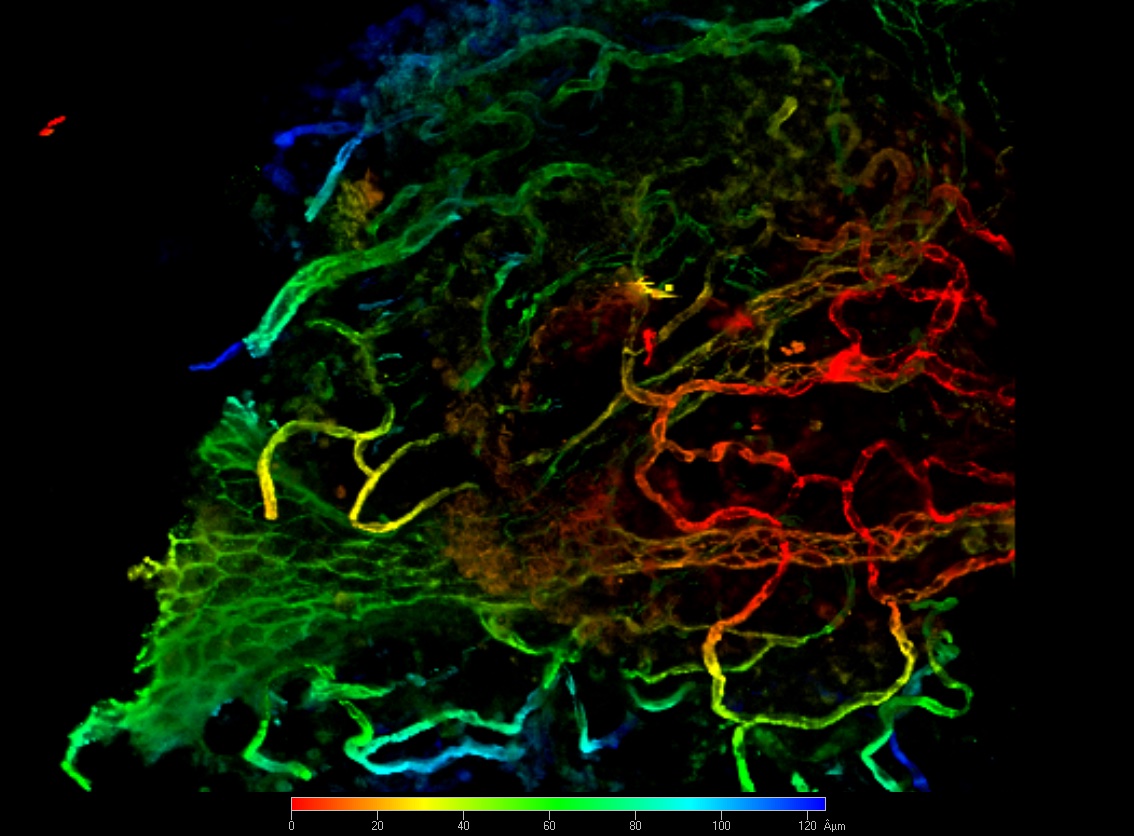
Tumor cell-derived extracellular matrix in tumor angiogenesis
Extracellular matrix (ECM) in tumors is recognized as an important driver of cancer cell aggressiveness. On the other hand, it is less known how tumor-derived extracellular matrix mechanically influences angiogenesis, tumor growth and dissemination. Using a syngeneic model of melanoma cells, we study how endothelial cells respond to the changes and remodeling of the tumoral ECM in vitro and in vivo. Remodeled ECM modulates intracellular mechano-trasducion signaling which in turn modulates the metabolic behavior both of tumor and endothelial cells. To this, label-free and high resolution fluorescence microscopies are used to visualize the 3D structure and remodeling of the ECM in vitro and in vivo systems and to study how the ECM remodeling affects intracellular mechano- and biochemical- signal transduction (e.g. mechanical forces across focal adhesion plaques, ROS production, Calcium fluxes, diffusion of transmembrane proteins along cell membrane). Also, FRAP/FRET/FLIM-FRET technologies are used to characetrize protein-protein interactions and protein dynamics.
VEGFR2 in ovarian cancer
Despite being considered an oncogene, VEGFR2 plays an anti-tumoral role in certain tumors. We employ cellular and in vivo models to investigate the biochemical and molecular bases of this unexpected role of VEGFR2 in high-grade serous ovarian carcinoma, a very aggressive cancer that is difficult to cure and often relapses. In particular we are focused on the role of VEGFR2 in the regulation of cell mechanotransduction.
Membrane dynamics of receptor tyrosine kinases
Membrane dynamics have emerged as crucial determinants of the function of cell surface receptors. In our lab we are interested in investigating how the dynamics of surface receptors along the membrane are intertwined with their function. Our studies are focused on the dynamics of tyrosine kinase receptors such as VEGFRs and FGFRs. Read references 2017, 2020, 2020.
Bio-scaffolds for the regeneration of brown adipose tissue
As the proportions of obesity increase worldwide, it becomes crucial to develop innovative and effective strategies to treat patients with obesity. The strategy we are pursuing consists in promoting adipose tissue health and browning through the implantation of bio-scaffolds. In this framework, angiogenesis plays a central role allowing for physiological adipose tissue expansion and preventing its dysfunction. In our lab we are studying novel natural bio-scaffolds that could support angiogenesis in beige adipose tissue.
Gremlin-1 in development, angiogenesis, cancer and metabolic disorders
Our group has contributed to the identification and characterization of Gremlin-1 as a pro-angiogenic ligand of VEGFR2. In our lab we are interested in studying the biochemical and molecular bases of gremlin-1 physio-pathological function. Considering the pathogenetic role of gremlin-1 in various human disorders, a better understanding of its function is of pivotal importance to design novel strategies to target gremlin-1 and its function in human pathologies. Read references 2007, 2008, 2010, 2012, 2015, 2016, 2023.